Rat Interleukin-1 beta Recombinant
Category: Recombinant Rat Cytokines$70.00 – $4,700.00
Description
Accession
Q63264
Source
Optimized DNA sequence encoding Rat Interleukin-1 beta mature chain was expressed in Escherichia Coli.
Molecular weight
Mature Rat IL-1 beta, is generated by the proteolytic removal of the signal peptide and propeptide.The molecule has a calculated molecular mass of approximately17 kDa. Recombinant rat IL-1 beta is a monomer protein consisting of 153 amino acid residue subunits. Recombinant rat IL-1 beta migrates as an approximately 17 kDa protein under non-reducing and reducing conditions in SDS-PAGE.
Purity
>98%, as determined by SDS-PAGE and HPLC
Biological Activity
The ED(50) was determined by thedose-dependent stimulation of thymidine uptake by murine D10S cells is ≤0.2 ng/ml, corresponding to a specific activity of ≥5 x107 units/mg.
Protein Sequence
MATVPELNCE IAAFDSEEND LFFEADRPQK IKDCFQALDL GCPDESIQLQ ISQQHLDKSF RKAVSLIVAV EKLWQLPMSC PWSFQDEDPS TFFSFIFEEE PVLCDSWDDD DLLVCDVPIR QLHCRLRDEQ QKCLVLSDPC ELKALHLNGQ NISQQVVFSM SFVQGETSND KIPVALGLKG LNLYLSCVMK DGTPTLQLES VDPKQYPKKK MEKRFVFNKI EVKTKVEFES AQFPNWYIST SQAEHRPVFL GNSNGRDIVD FTMEPVSS
Endotoxin
Endotoxin content was assayed using a LAL gel clot method. Endotoxin level was found to be less than 0.1 ng/µg(1EU/µg).
Presentation
Rat IL-1 beta was lyophilized from a 0.2 μm filtered solution in PBS, pH.
Reconstitution
A quick spin of the vial followed by reconstitution in distilled water to a concentration not less than 0.1 mg/mL. This solution can then be diluted into other buffers.
Storage
The lyophilized protein is stable for at least years from date of receipt at -20° C. Upon reconstitution, this cytokine can be stored in working aliquots at2° -8° C for one month, or at -20° C for six months, with a carrier protein without detectable loss of activity. Avoid repeated freeze/thaw cycles.
Usage
This cytokine product is for research purposes only.It may not be used for therapeutics or diagnostic purposes.
Biological Process
Molecular function
Molecular function
Molecular function
Methods
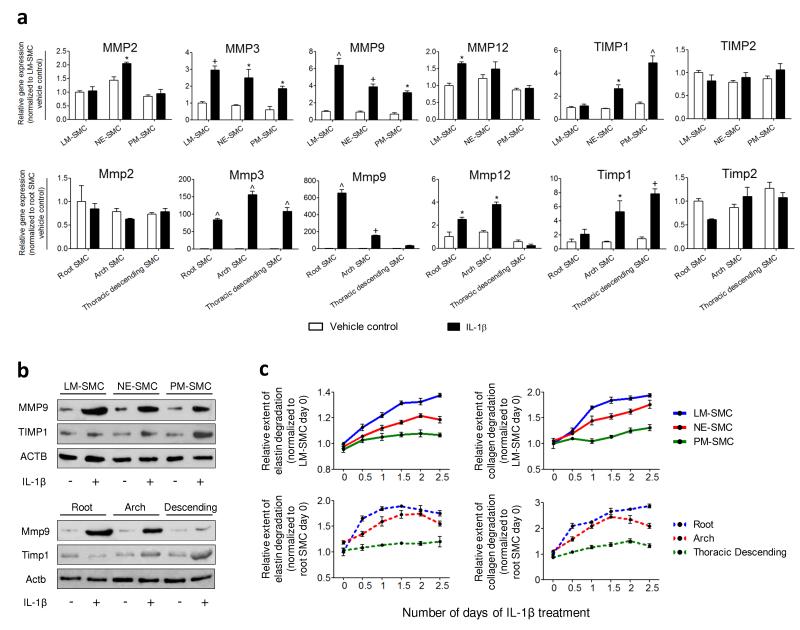
HPSC-derived SMC subtypes predict MMP and TIMP expression and activity in rat aortic SMCs of corresponding origins.
- The origin-specific SMCs (top panel) replicated similar trends of elastin and collagen degradation as the rat aortic SMCs (bottom panel) in response to IL-1β.
Proteolytic assays
- 96-well plates were coated with either 0.1 mg/ml of elastin or collagen type I, of which both were FITC conjugated .
- HESC-derived SMCs or rat SMCs were then seeded 6,000 cells/well.
- Following incubation overnight, 10 ng/ml of IL-1β was added in serum-free media.
- Activity of secreted proteases was assessed by the increase of fluorescence measured at excitation/emission = 490 nm/ 520 nm using the Wallac Envision 2104 multilabel reader .
- To take into account any differential cell proliferation due to IL-1β, the fluorescence readout was normalized against the corresponding cell number/well.
Preparation of bone marrow-derived mast cells
- Bone marrow cells were harvested from femurs of LEW rats and cultured in RPMI 1640 supplemented with 10% FBS, 2 mM L-glutamine, 1 mM sodium pyruvate, 1 mM HEPES, 50 µM 2-mercaptoethanol, 100 U/ml penicillin, and 100 µg/ml streptomycin.
- The cells were cultured in the presence of 10 ng/ml of recombinant rat IL-3β , and the nonadherent cells were passaged every 3 days.
- 4 weeks later, the cells were used as mast cells for experiments and referred to as BMMCs
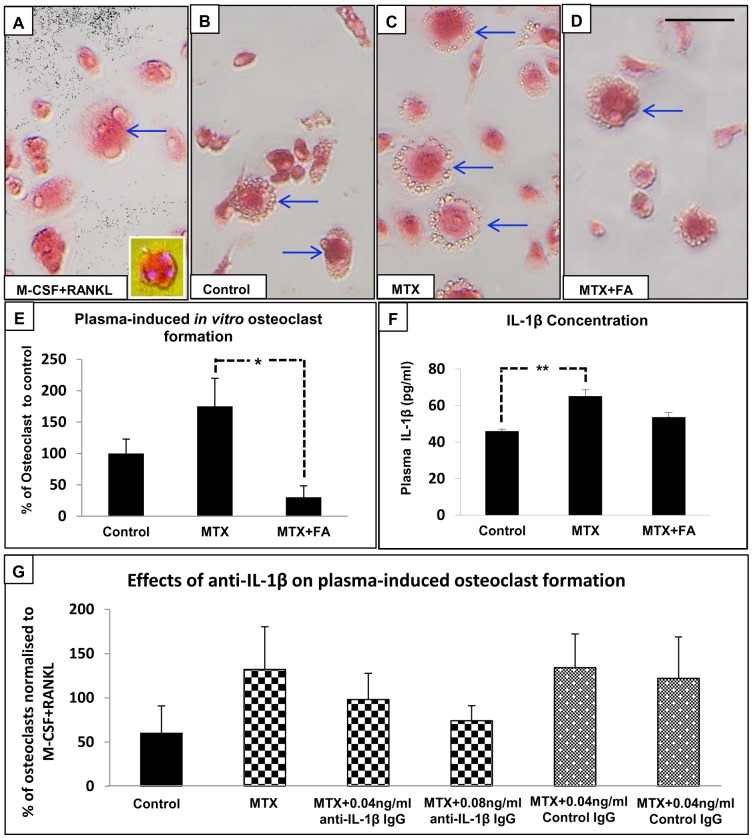
Osteoclast formation in vitro as induced by plasma from rats treated with MTX alone or with supplementary folinic acid (FA) for 6 weeks.
- Plasma concentrations of IL-1β at week 6.
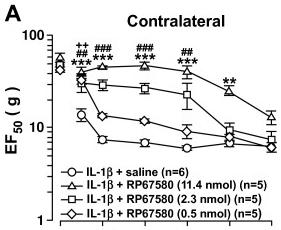
Inhibition of NK1-R in the RVM attenuates IL-1β-induced bilateral hyperalgesia.
- IL-1β (160 fmol/0.5 μl) was microinjected unilaterally into the Vi/Vc 15 min prior to RP67580 and L-733,060 microinjection into the RVM (0.5 μl).
Surgical preparation/cannulation
- Rats were anesthetized using 50 mg/kg of pentobarbital sodium (i.p) and 2-3% isoflurane inhalation in a 30/70% oxygen/nitrogen gas mixture.
- Rats were placed in a stereotaxic device ( , 900).
- A midline incision was made in the scalp after debridement and sterilization of the surgical field with iodine wash. For administration of drugs via microinjection, guide cannulae (C315G, 26 gauge , , ) were implanted and cemented into the skull.
- A midline opening was made in the skull using a dental drill and a guide cannula was lowered into the ventral Vi/Vc transition zone or RVM by referring to the rat brain atlas [3 receptor antagonist Y-25130 (Tocris; , ) were microinjected into the Vi/Vc transition zone.
- The Neurokinin-1 receptor antagonists L-733,060 and RP67580 were microinjected into the RVM.
- IL-1β, IL-1 receptor antagonist, Y-25130, and L-733,060 were reconstituted in deionized water while RP67580 was reconstituted in DMSO.
Transwell Migration Assay-in vitro Blocking Experiments
-
In vitro motility assays were performed as previously described 4/100 µL density to the insert of a transwell plate with 8
µ m pore size in EBM-2: EGM-2 (4∶1) medium. - Cells were then allowed to migrate for 6 h across the pore membrane, toward medium'>Sham medium or SS-AF-MSC-CM.
- After the 6 h incubation period, the non-migrated cells were removed from the top of the insert with a wet cotton swab.
- The migrated cells were then fixed with 4% (w/v) paraformaldehyde on the membrane and stained sequentially with eosin and hematoxilin (all from .).
- Migration was quantified by counting the nuclei that passed through the filter.
- Photographs of the stained nuclei were taken from a minimum of 10 fields of view (20Χ) for each membrane using an inverted TE300 microscope fitted with a cooled CCD camera and Simple PCI software and experiments were performed repeated twice.
- Statistical analysis was…
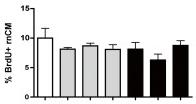
Conditioned medium of ADSC promotes proliferation rate of cardiomyocytes.
- This proliferation rate was not significantly influenced by treatment with TNF-α or IL-1β.
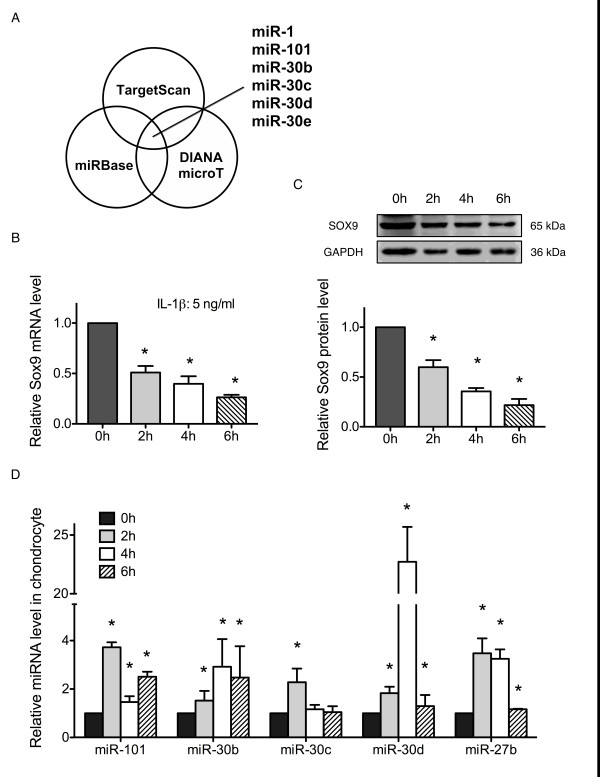
microRNA prediction and its expression in IL-1β-treated chondrocytes.
- Primary rat chondrocytes were treated with IL-1β (5 ng/ml) for 2, 4 and 6 h. Sox9 expression was analyzed by real-time PCR and western blot.
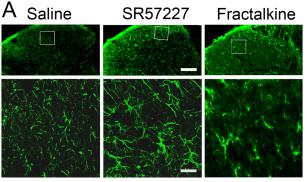
Upregulation of IL-1β in the dorsal horn astrocytes and its involvement in pain behavior after intrathecal injection of SR57227.
- A. SR 57227 (10 pmol, i.t., n = 4) or fractalkine (40 ng, i.t., n = 4) produced an increased expression of IL-1β (lower panels) in the area from the inset in the spinal dorsal horn (upper panels) compared to saline at 2 h or 1 h after injection, respectively.