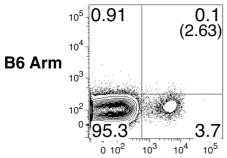
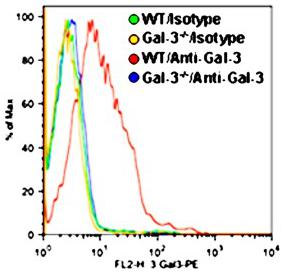
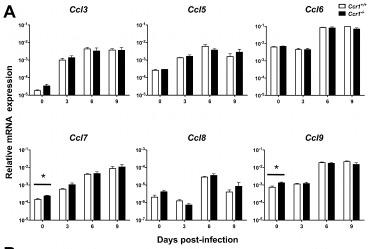
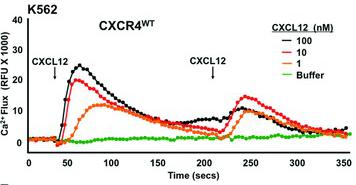
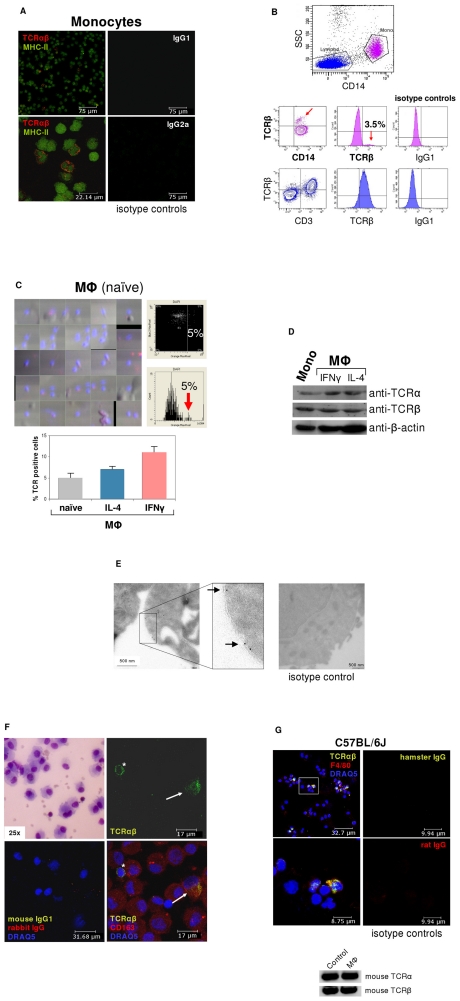
Mouse TNF-alpha Recombinant
Categories: Recombinant Mouse CytokinesTNF family$70.00 – $2,700.00
Description
Accession
P06804
Source
Optimized DNA sequence encoding Mouse Tumor Necrosis Factor-alpha extracellular domain was expressed in Escherichia Coli.
Molecular weight
Native Mouse Tumor Necrosis Factor-alpha is generated by the proteolytic removal of the signal peptide and propeptide, this molecule has a calculated mass of approximately 17kDa. Recombinant Mouse TNF is a monomer protein,consisting of 157 amino acids and migrates as an approximately17 kDa protein under reducing conditions.
Purity
>97%, as determined by SDS-PAGE and HPLC
Biological Activity
The ED(50) was determinedcytolysis of murine L929 cells in the presence of Actinomycin D is ≤.05 ng/ml, corresponding to a specific activity of ≥3 x units/mg.
Protein Sequence
MSTESMIRDV ELAEEALPQK MGGFQNSRRC LCLSLFSFLL VAGATTLFCL LNFGVIGPQR DEKFPNGLPL ISSMAQTLTL RSSSQNSSDK PVAHVVANHQ VEEQLEWLSQ RANALLANGM DLKDNQLVVP ADGLYLVYSQ VLFKGQGCPD YVLLTHTVSR FAISYQEKVN LLSAVKSPCP KDTPEGAELK PWYEPIYLGG VFQLEKGDQL SAEVNLPKYL DFAESGQVYF GVIAL
Endotoxin
Endotoxin content was assayed using a LAL gel clot method. Endotoxin level was found to be less than 0.1 ng/µg(1EU/µg).
Presentation
Recombinant mouse TNF alpha was lyophilized from.2 μm filtered PBS solution, pH7.0.
Reconstitution
A quick spin of the vial followed by reconstitution in distilled water to a concentration not less than 0.1 mg/mL. This solution can then be diluted into other buffers.
Storage
The lyophilized protein is stable for at least years from date of receipt at -20° C. Upon reconstitution, this cytokine can be stored in working aliquots at2° -8° C for one month, or at -20° C for six months, with a carrier protein without detectable loss of activity. Avoid repeated freeze/thaw cycles.
Usage
This cytokine product is for research purposes only.It may not be used for therapeutics or diagnostic purposes.
Interactor
P34015
Molecular function
Methods
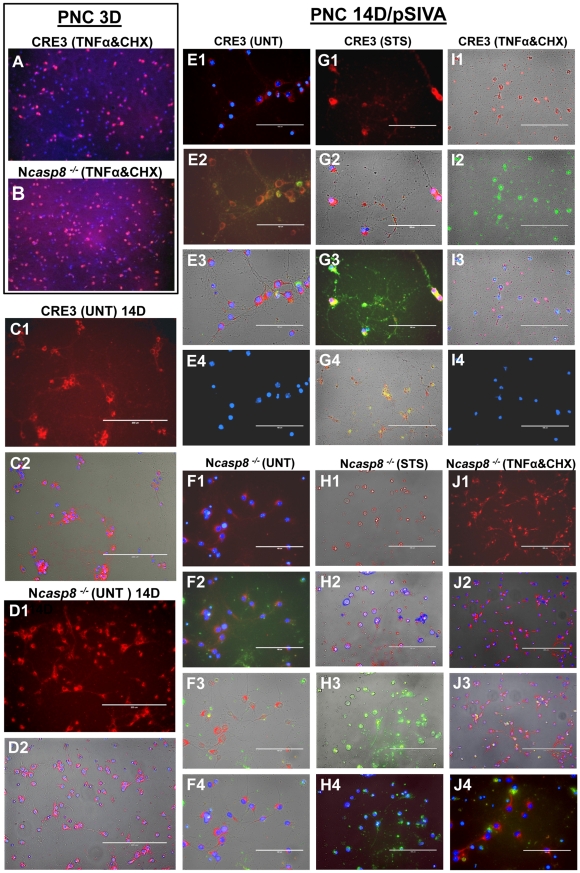
Caspase 8 gene ablation provides neuroprotection in vitro in primary neuronal cultures and brain organotypic cultures.
- 3-day-old PNC derived from embryos (E16.5) of CRE3 and Ncasp8
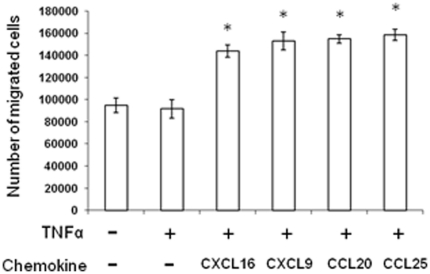
Transendothelial migration of mMSCs in response to chemokines.
- MAECs were treated with or without TNFα and in the presence or absence of CXCL16, CXCL9, CCL20 and CCL25 (each at 10 ng/ml).
Materials
- Tetramethylammonium hydroxide was from Shanghai Lingfeng Chemical Reagent Co Ltd, Shanghai, China.
- Other materials were RPMI 1640 medium, penicillin, and streptomycin , fetal bovine serum (, , , the ), recombinant murine granulocyte-macrophage colony-stimulating factor, recombinant murine interleukin-4, CD11c, B7-1 (CD80), B7-2(CD86), MHC II, and chemokine receptor 7 (eBioscience, San Diego, CA), tumor necrosis factor-α, interleukin-1β, and interleukin-6 , prostaglandin E2 , a Prussian blue staining kit .
- Annexin V and propidium iodide , and the Cell Counting Kit-8 .
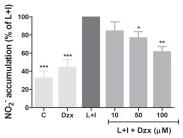
Anti-inflammatory effects of diazoxide pre-treatment in microglial cell cultures stimulated with LPS and IFNγ.
- Nitrite accumulation , and TNF-α and IL-6 release in control (unstimulated cells), DZX (unstimulated cells pretreated with 100 μM diazoxide), diazoxide and LPS/IFNγ + Diazoxide (10 μM to 100 μM) normalized for LPS/IFNγ untreated cells.
Flow cytometric analysis of endothelial VCAM-1 and E-selectin
- Human umbilical vein endothelial cells were cultured in EGM-2 according to the manufacturer’s protocol.
- HUVEC (passage 3–5) were plated in 24-well plates with 1×105 cells/400 µl per well in EGM-2 plus 20% FBS.
- After a 5 hour re-attachment period, cells were pre-incubated for 16 hours with HDL isolated from pooled serum samples (n=6), after which TNF-α (0.5 ng/ml) was added to the culture medium for an additional 4 hours.
- Cell-surface expression of VCAM-1 and E-selectin were then measured by flow cytometry, using a VCAM-1 monoclonal antibody followed by an anti-mouse FITC conjugated antibody , or an E-selectin PE conjugated antibody .
- Cells were detached using 5 mM EDTA in PBS.
- Cellular FITC and PE were then analyzed by flow cytometry (C6 Flow Cytometer, Accuri Cytometers ).
- Controls included an isotype-matched control antibody and no primary antibody.
- Native HDL3 (isolated by sequential ultracentrifugation of human plasma) at concentration of 0.5 mg/ml…
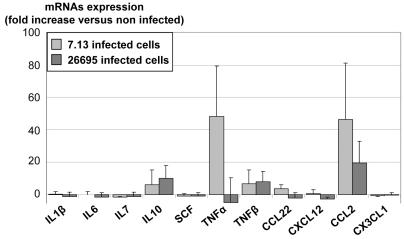
Epithelial cytokine expression and MSC migration.
- B/ Supernatants from non-infected (white bars), 7.13- (light grey bars) or 26695- (dark grey bars) infected cells were assessed for TNFα and CCL2 expression by ELISA.
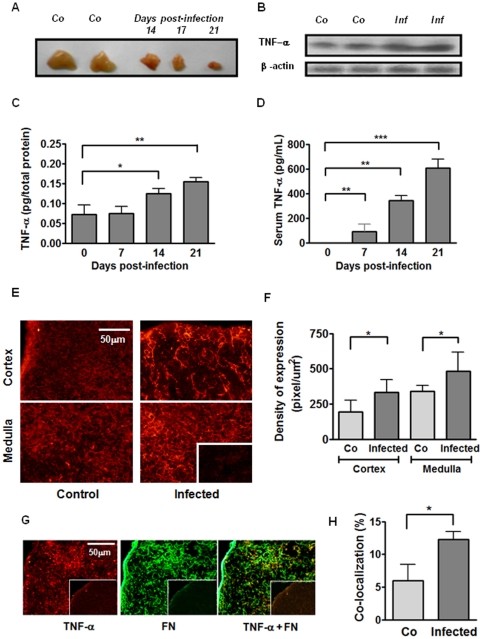
Enhanced circulating and intrathymic contents of TNF-α parallel the thymic atrophy in T. cruzi acutely-infected mice.
- A) Representative picture showing two thymuses from healthy control mice (Co) and the progressive thymic atrophy after 14, 17 and 21 days of acute infection; B) TNF-α protein detection in thymus homogenates by western blot.
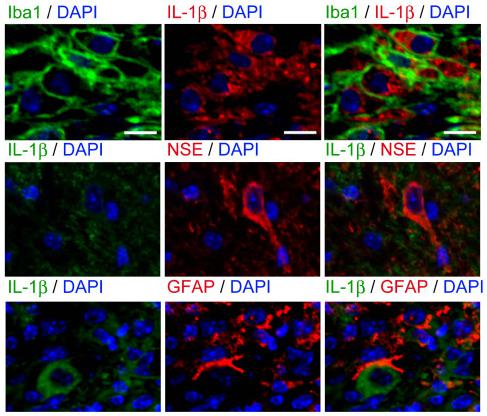
Less proinflammatory cytokine induction was evident in IL-1 KO mice after SCI.
- TNFα level in the spinal cord of the wild-type (open squares and solid line) and IL-1 KO mice (closed square and dotted line).
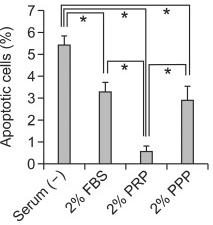
PRP decreased the apoptotic cell death and caspase 3 activity induced by TNF-α and cycloheximide in ccdPAs.
- The cells were treated with TNF-α and cycloheximide for 5, 30, 60, and 120 min in the medium with 2% FBS (closed bar) or 2% PRP (open bar).
Transendothelial Migration and Invadopodia (in Vitro Filipodia) Formation Assay of the T Lymphoma Cells
- Trans-endothelial migration of T-cells was measured as described 6 cells) were seeded on the upper compartment on top of the endothelial monolayer in 100 µl.
- After 16 hours of treatment, the tissue culture wells were removed and the lower compartment cells were collected to determine the number of transmigrated lymphocytes.
- To assess the formation of leading edge filipodia, the T lymphoma cells were labelled with cell tracker green and added to confluent monolayers of tumour necrosis factor α (TNF-α) (cat 300-01A, , , ) activated bEnd.3 cells grown on glass cover slips that had been coated with 2 µg/ml fibronectin.
- After, 16 hours of incubation, non adherent cells to the cover slips were removed by gentle washing the adherent cells were fixed with 4% paraformaldehyde and stained with DAPI and TRITC-phalloidin.
- Z stack images were captured using Axiovision 4.1 Zeiss microscope and analyzed.