Mouse Stem Cell Factor Recombinant
Categories: PDGF familyPDGF familyRecombinant Mouse Cytokines$160.00 – $3,700.00
Description
Accession
P20826
Source
Optimized DNA sequence encoding Mouse SCF mature chain was expressed in Insect cells.
Molecular weight
Native Mouse Stem Cell Factor is generated by the proteolytic removal of the signal peptide and propeptide, the molecule has a calculated molecular mass of approximately 18 kDa. Recombinant SCF is a monomer protein consisting of 165 amino acid residue subunits, and due to glycosylation migrates as an approximately 18 kDa protein under reducing conditions in SDS-PAGE.
Purity
>95%, as determined by SDS-PAGE and HPLC
Biological Activity
The ED(50) was determined by the dose-dependent stimulation of theproliferation of human TF-1 cells is <2.0 ng/ml, corresponding to a specific activity of > x units/mg.
Protein Sequence
MKKTQTWIIT CIYLQLLLFN PLVKTKEICG NPVTDNVKDI TKLVANLPND YMITLNYVAG MDVLPSHCWL RDMVIQLSLS LTTLLDKFSN ISEGLSNYSI IDKLGKIVDD LVLCMEENAP KNIKESPKRP ETRSFTPEEF FSIFNRSIDA FKDFMVASDT SDCVLSSTLG PEKDSRVSVT KPFMLPPVA A SSLRNDSSSS NRKAAKAPED SGLQWTAMAL PALISLVIGF AFGALYWKKK QSSLTRAVEN IQINEEDNEI SMLQQKEREF QEV
Endotoxin
Endotoxin content was assayed using a LAL gel clot method. Endotoxin level was found to be less than 0.1 ng/µg(1EU/µg).
Presentation
Recombinant Stem cell factor was lyophilized from a 0.2 μm filtered solution inmM Tris,% trehalose, pH.5.
Reconstitution
A quick spin of the vial followed by reconstitution in distilled water to a concentration not less than 0.1 mg/mL. This solution can then be diluted into other buffers.
Storage
The lyophilized protein is stable for at least years from date of receipt at -20° C. Upon reconstitution, this cytokine can be stored in working aliquots at2° -8° C for one month, or at -20° C for six months, with a carrier protein without detectable loss of activity. Avoid repeated freeze/thaw cycles.
Usage
This cytokine product is for research purposes only.It may not be used for therapeutics or diagnostic purposes.
Biological Process
Molecular function
Methods
Cell culture
- Peripheral blood mobilized, purified human CD34+ cells were obtained from the Hematopoietic Cell Processing Core at the Fred Hutchinson Cancer Center as previously described
Generation of BMDC and BMMC
- For BMMC induction, 1×106 BM cells were cultured supplemented with 5 ng/ml recombinant murine SCF and IL-3 for more than three weeks (>98% expressed c-kit and FcεRIα).
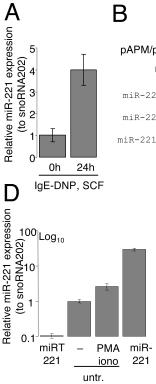
MiR-221 is upregulated upon mast cell activation and its expression levels can be altered using lentivirus-based systems.
- A) Differentiated BMMCs were either left resting or were stimulated with 1.5 µg/mL IgE anti-DNP, 0.2 µg/mL DNP-HSA and 20ng/mL SCF prior analysis of miR-221 expression by TaqMan qRT-PCR.
Mast cell culture from bone marrow
- Mouse bone marrow-derived cultured mast cells (BMCMC) were differentiated from femoral bone marrow by culture in medium supplemented with recombinant mouse IL-3 and stem cell factor(rmSCF, , ) as described −/− bone marrow showed similar granular morphology, levels of active tryptase, and expression of FcεRIα and CD117, indicating that they mature similarly when cultured in the presence of IL-3 and SCF.
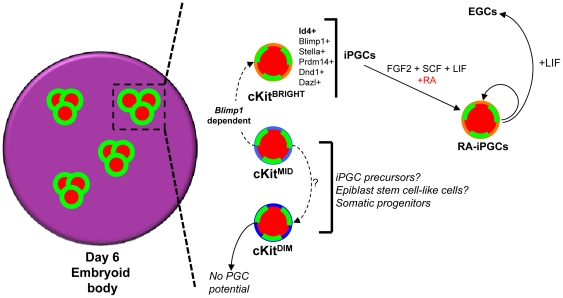
Model for iPGC emergence from SSEA1/Oct4+ clusters in EBs.
- Using a differential colony forming assay in the presence FGF2, SCF, LIF and RA which promotes survival and proliferation of PGCs, we show that RA-iPGC potential is highest in the cKitbright fraction and is absent in the cKitdim subpopulation of SSEA1+ cells.
Expansion of CD34+ cells in serum free medium
- Isolated CD34+ cells were seeded at a density of 5×104cells per 500 µl of medium'>Stempro medium , in 24-well tissue culture plates .
- Growth factors used were IL-6, SCF, TPO, and Flt-3-L at a final concentration of 25 ng/ml with (test cells) and without (control cells) the addition of either zVADfmk −100 nM or zLLYfmk −10 µM (MP, , ).
- After the 10th day of culture, the cells were collected from the suspension culture, and centrifuged (1000 rpm for 5 minutes).
- The cells were used for assessing the
in vitro homing properties or were used forin vivo homing studies in NOD/SCID mice.
In vitro culture of Eos and MDSCs
- For obtaining Eos, naïve bone marrow cells were cultured in 10% medium'>FBS/PMI medium supplemented with stem cell factor and FLT3-ligand for 4 days, and then with medium containing recombinant mouse IL-5 .
- On day 12, the cells were collected for May-Grünwald-Giemsa staining and flow cytometry analysis6 cells per ml in 10% FBS/RPMI medium, supplemented with 40 ng ml−1 GM-CSF.
- On day 4, the cells were collected for FACS analysis of CD11b+ CD11c− Ly-6C+ Ly-6Glow MDSCs
ES cell lines and EB differentiation
- Two days prior the onset of differentiation, ES cells were transferred to Iscove's Modified Dulbecco's Medium (IMDM,) containing the above components.
- EBs in suspension culture were then generated as described