Human TGF Beta-1 Recombinant
Categories: CytokinesRecombinant Human Cytokines$70.00 – $3,500.00
Description
Accession
P01137
Source
Optimized DNA sequence encoding Human Transforming Growth Factor Beta-1 mature chain was expressed in HEK293 cells
Molecular weight
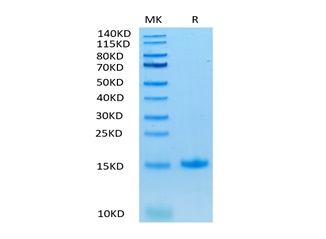
Native human TGF-b1, generated by the proteolytic removal of the signal peptide and propeptide,the molecule has a calculated molecular mass of approximately 26 kDa. Recombinant TGF-beta 1is a disulfide-linked homodimer protein consisting of 2x113 amino acid residue subunits. TGF-beta 1 migrates as an approximately 12 - 13 kDa protein under reducing conditions and as 26kDa under nonreducing conditions .
Purity
>95%, as determined by SDS-PAGE and HPLC.
Biological activity
The ED(50) was determined by the ability to inhibit the proliferation of mink lung epithelial cells 0.05-0.1 ng/ml, corresponding to a specific activity of 2x 107 units/mg.
Endotoxin
Endotoxin content was assayed using a LAL gel clot method. Endotoxin level was found to be less than 0.1 ng/µg(1EU/µg).
Presentation
Recombinant TGF-b1 is lyophilized from 0.2 μm filtered PBS solution pH 7.4.
Reconstitution
A quick spin of the vial followed by reconstitution in distilled water to a concentration not less than 0.1 mg/mL. This solution can then be diluted into other buffers.
Storage
Upon reconstitution, this cytokine can be stored in working aliquots at 2° - 8° C for one month, or at -20° C for six months. Avoid repeated freeze/thaw cycles.
Usage
This cytokine product is for research purposes only.It may not be used for therapeutics or diagnostic purposes.
Interactor
Interactor
Interactor
P18564
Interactor
Interactor
Interactor
Q03167
Interactor
Interactor
Q14766
Interactor
Molecular function
Molecular function
Methods
Induction of Chondrogenesis
- MSC from passage 3 were harvested with trypsin/EDTA and pellets consisting of 5×105 cells were formed by centrifugation.
- Chondrogenic induction medium consisted of DMEM high glucose supplemented with 0.1 µM dexamethasone, 0.17 mM ascorbic acid 2-phosphate, 5 µg/ml transferrin, 5 ng/ml selenous acid, 1 mM sodium pyruvate, 0.35 mM proline, 1.25 mg/ml BSA, 100 units/ml penicillin, 100 µg/ml streptomycin , 5 µg/ml insulin and 10 ng/ml TGF-β1 .
- Pellets were cultured for 6 weeks in medium'>medium'>chondrogenic medium'>medium'>induction medium'>medium and medium'>medium was changed three times the weeks.
Experimental Induction of Ectopic Digits and CCN Treatments
- Ectopic digits were induced by local implantation of heparin beads incubated for 1 hr in 2 µgr/ml rh-TGFβ1 .
- For this purpose eggs were windowed at 5.5 id and the bead (raging between 80 and 150 µm of diameter) was implanted in the third interdigit of the right leg bud.
- The contralateral left limb or limbs treated with beads incubated in PBS, were employed as controls.
- After manipulation the eggs were sealed and further incubated until processing.
Adsorption of growth factors to osteoblast-secreted ECMs
- Recombinant human TGF-β1 and BMP-2 were reconstituted to a final concentration of 1 mg/ml in PBS containing bovine serum albumin (BSA) as a carrier.
- The growth factors were fluorescently labeled using the Dylight 488 labeling kit as described by the manufacturer.
- Briefly, the growth factors were incubated with the fluorescent dye for 1 h at room temperature.
- The unincorporated dye was removed by passing the mix through a desalting spin column.
- The labeled growth factors were diluted in PBS to obtain a range of concentrations from 0 to 200 ng/ml.
- ECMs prepared as described above were incubated overnight at 4°C with 200 µl of PBS containing either TGF-β1 or BMP-2 at different concentrations.
- After 24 h, the growth factor solution was collected, ECMs were rinsed twice with PBS to remove unbound growth factor, and the distribution of bound protein on ECMs was imaged using a Nikon Eclipse TE2000-U fluorescent microscope.
- Growth factor-bound ECMs…
2.4. Chondrogenic Differentiation of cASCs
- For chondrogenic induction, the micromass culture technique was used [2 flasks (with flat cultivation surface) using chondrogenic induction medium at a concentration of 2 × 106/flask.
- Chondrogenic induction medium consisted of DMEM, 10% FCS, 0.1
M Dexa, 50 M Asc-2p, 0.35 g/100mL D(+)-glucose (# G-7021, Sigma-Aldrich Chemie), 50 mg/mL (1x) ITS + 1 liquid media supplement (10 g/mL insulin, 5.5 g/mL transferrin, 5 ng/mL selenium, 0.5 mg/mL bovine albumin, 4.7 g/mL linoleic acid) (# I-2521, Sigma-Aldrich Chemie), and 10 ng/mL transforming growth factor (rhTGF- β 1; #100-21, PeproTech GmbH, Hamburg, Germany). - After seeding, cells were allowed to adhere for 4 hours (37°C, humidified atmosphere, 5% CO2).
- Afterwards, the cells were maintained in induction medium for 21 days.
- The culture medium was changed every third day.
- Negative controls consisted of cASCs maintained in noninductive basal medium.
- Fixation of the cells was carried out as described above.
- Chondrogenic induction was confirmed by Alcian blue staining for chondrocyte-specific proteoglycans.
Activation of MAs and Early DC Differentiation
-
For DC differentiation, following early Mo–MA maturation for 12 h, the cells were cultured with 500 U/ml IL-4+800 U/ml GM-CSF for 120 h as described, or the cytokine cocktail was supplemented with TGF-
β in various concentrations. - Alternatively, DCs were treated with rosiglitazone as published previously. At the indicated time points, cells were collected for paraffin embedding and IF staining, or for qRT-PCR.
- Alternatively, the cultured cell monolayers were stained with Oil-Red-O using standard methods.
- For combination of the lipid staining with IHC, following CPM-IF staining using FITC fluorochrome, monolayers were treated with 50 ng/ml Nile-red , washed and counterstained with DAPI.
Cell culture and siRNA transfection
- Human mammary epithelial cells infected with a retrovirus carrying hTERT and the oncogenic H-RasV12 (-TR) allele were provided by R. A.
Cell culture and immunocytochemical staining
- The NIH 3T3 fibroblast cell line and HK2 cell line from ATCC were cultured in medium'>DMEM medium supplemented with 10% FBS until the cells were 80% confluent.
- Cells were then incubated in medium'>DMEM medium containing 0.2% FBS for 24 hr.
- NIH3T3 cells and HK2 cells were cultured for 24 to 48 hr in the presence of 10 ng/ml recombinant human TGF-β1 or 5 µg/ml aristolochic acid , with or without addition of 50 nmol rapamycin (LC lab, , ) for 12 hr.
- Portions of NIH3T3 cells and HK2 cells were cultured in four-chamber glass and immunocytochemical staining was performed as previously described
Osteoclast differentiation and pit formation assays
- To test the effect of dasatinib on the bone resorption capacity of medium'>OCs, PBMCs at 0.6×106 cells/well were seeded on calcium-coated slides in 200 µl of medium'>OC medium supplemented with 5 ng/mL TGF-β1 and 1 µM dexamethasone for 17 days
Fibroblast differentiation assay
- Confluent cultures of Human MRC-5 fibroblast line were serum-starved for 48 hours, incubated with(20 or 40 µM) or vehicle (DMSO), then treated with 10 ng/ml TGF-β1 or Phosphate-Buffered Saline (PBS) vehicle for 72 hours