Human Neuregulin 1 beta Recombinant
Categories: EGF familyEGF familyRecombinant Human Cytokines$70.00 – $1,350.00
Description
Accession
Q02297
Source
Optimized DNA sequence encoding Human Neuregulin-1 beta EGF domain was expressed in Escherichia Coli.
Molecular weight
Recombinant human Neuregulin-1 beta EGF domain is a monomeric protein consisting of 62 amino acid residue subunits, and migrates as an approximately 7 kDa protein under non-reducing and reducing conditions in SDS-PAGE.
Purity
>96%, as determined by SDS-PAGE and HPLC
Biological Activity
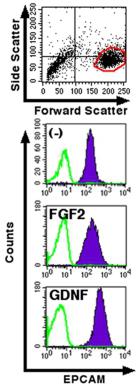
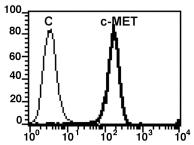
The ED(50) was determined by the dose-dependent proliferation of human MCF-7 cells was found to be <0.3 ng/ml, corresponding to a specific activity of 3x107 Units/mg.
Protein Sequence
MSERKEGRGK GKGKKKERGS GKKPESAAGS QSPALPPRLK EMKSQESAAG SKLVLRCETS SEYSSLRFKW FKNGNELNRK NKPQNIKIQK KPGKSELRIN KASLADSGEY MCKVISKLGN DSASANITIV ESNEIITGMP ASTEGAYVSS ESPIRISVST EGANTSSSTS TSTTGTSHLV KCAEKEKTFC VNGGECFMVK DLSNPSRYLC KCPNEFTGDR CQNYVMASFY KHLGIEF MEQ KRVLTITGIC IALLVVGIMC VVAYCKTKKQ RKKLHDRLRQ SLRSERNNMM NIANGPHHPN
Endotoxin
Endotoxin content was assayed using a LAL gel clot method. Endotoxin level was found to be less than 0.1 ng/µg(1EU/µg).
Presentation
Recombinant Neuregulin-1 was lyophilized from a 0.2 μm filtered 10mM PB, pH7.0, containing %5 mannitol.
Reconstitution
A quick spin of the vial followed by reconstitution in distilled water to a concentration not less than 0.1 mg/mL. This solution can then be diluted into other buffers.
Storage
The lyophilized protein is stable for at least years from date of receipt at -20° C. Upon reconstitution, this cytokine can be stored in working aliquots at 42° -8° C for one month, or at -20° C for six months, with a carrier protein without detectable loss of activity. Avoid repeated freeze/thaw cycles.
Usage
This cytokine product is for research purposes only.It may not be used for therapeutics or diagnostic purposes.
Interactor
Molecular function
Methods
Differentiation to a Schwann cell phenotype
- Human ADSCs were induced into neurospheres.
- Briefly, we harvested human ADSCs (80–90% confluence) and then plated them in plastic dish a concentration of 1–2 × 105/cm2 in DMEM: F12 supplemented with 20 ng/ mL EGF , 20 ng/mL basic fibroblast growth factor (bFGF) and 2% B27 (1:50) at 37°C in 5%CO2.
- We added fresh medium every 3 to 4 days.
- After 7 days, neurospheres were triturated using a fire-polished Pasteur pipette and re-plated in Laminin coated six-well chamber slides contain DMEM: F12 supplemented with 10% FBS, 14 μM forskolin , 5 ng/mL platelet-derived growth factor-AA (PDGF, ), 10 ng/mL bFGF and 200 ng/mL recombinant human heregulin-beta1 (HRG) for terminal differentiation.
- The cells were incubated for 9 days under these conditions, and then harvested for investigation.
Differentiation to a Schwann cell phenotype
- Human ADSCs were induced into neurospheres.
- Briefly, we harvested human ADSCs (80–90% confluence) and then plated them in plastic dish a concentration of 1–2 × 105/cm2 in DMEM: F12 supplemented with 20 ng/ mL EGF , 20 ng/mL basic fibroblast growth factor (bFGF) and 2% B27 (1:50) at 37°C in 5%CO2.
- We added fresh medium every 3 to 4 days.
- After 7 days, neurospheres were triturated using a fire-polished Pasteur pipette and re-plated in Laminin coated six-well chamber slides contain DMEM: F12 supplemented with 10% FBS, 14 μM forskolin , 5 ng/mL platelet-derived growth factor-AA (PDGF, ), 10 ng/mL bFGF and 200 ng/mL recombinant human heregulin-beta1 (HRG) for terminal differentiation.
- The cells were incubated for 9 days under these conditions, and then harvested for investigation.
Differentiation of human AFS cells into a Schwann cell phenotype
- To initiate human AFS cells differentiation into a Schwann cell phenotype, AFS cells were dissociated (80–90% confluence) with 0.25% trypsin/EDTA and subsequently plated on 6 cm plastic dishes at a concentration of 105/cm2 in media consisting of α-MEM and 1 mM β-mercaptoethanol .
- After 24 hours, media was removed, cells were washed with PBS for 3 times, and media consisting of α-MEM, % Bovine Serum and 35 ng/ml retinoic acid was added.
- After 72 hours, cells were washed with PBS for 3 times and media was replaced with differentiation media consisting of α-MEM, 10% Fetal Bovine Serum , 20 ng/mL epidermal growth factor (EGF, ), 20 ng/mL basic fibroblast growth factor (bFGF, ), 5 mM forskolin , 5 ng/mL platelet-derived growth factor-AA (PDGF-AA, ) and 200 ng/mL recombinant human heregulin-beta1 (HRG, ).
- Media were additionally supplemented with 2.5 mM L-Glutamine, 50 mg/L…
Cell culture
- Undifferentiated CyT49 hESCs (ViaCyte, Inc. San iego CA) were maintained on EmbryoMAX Primary Mouse Embryo Fibroblasts (MEF) feeder layers in 10/10 media [MEM/F12 , 10% XenoFree KnockOut Serumeplacement , 1x MEM non-essential amino acids , 1x GlutaMAX , 1x penicillin/streptomycin (10,000 U/mL) , 10 nM β-mercaptoethanol , supplemented with 10 ng/mL Activin A and 10 ng/mL Heregulin-β1 ] 5 or 1×106 on 35 and 60 mm plates, respectively.
- Cells to be differentiated were plated on Growth Factor Reduced ( 1∶75 in DMEM/F12) coated plates.
- The cells derived in this study may be obtained upon written consent from ViaCyte Inc.