Human Interferon-alpha 2a Recombinant
Categories: Interferon-IL10 familyRecombinant Human Cytokines$70.00 – $880.00
Description
Accession
P01563
Source
Optimized DNA sequence encoding Human Interferon-alpha mature chain was expressed in Escherichia Coli.
Molecular weight
Recombinant human IFN-alphaa, generated by the proteolytic removal of the signal peptide and propeptide, and has a calculated molecular mass of approximately 19 kDa. Recombinant Interferon alphaais a monomeric protein consisting of 165 amino acid residue subunits, and migrates as an approximately 19 kDa protein under non-reducing conditions and reducing conditions in SDS-PAGE.
Purity
>98%, as determined by SDS-PAGE and HPLC
Biological Activity
Theactivity was determined bya viral resistance assay of Human WISH cells, andwas found to be in the range ofx108 IU/mg.
Protein Sequence
MALTFALLVA LLVLSCKSSC SVGCDLPQTH SLGSRRTLML LAQMRKISLF SCLKDRHDFG FPQEEFGNQF QKAETIPVLH EMIQQIFNLF STKDSSAAWD ETLLDKFYTE LYQQLNDLEA CVIQGVGVTE TPLMKEDSIL AVRKYFQRIT LYLKEKKYSP CAWEVVRAEI MRSFSLSTNL QESLRSKE
Endotoxin
Endotoxin content was assayed using a LAL gel clot method. Endotoxin level was found to be less than 0.1 ng/µg(1EU/µg).
Presentation
Recombinant Interferon alphaawas lyophilized from a 0.2 μm filtered PBS solution pH7.0.
Reconstitution
A quick spin of the vial followed by reconstitution in distilled water to a concentration not less than 0.1 mg/mL. This solution can then be diluted into other buffers.
Storage
The lyophilized protein is stable for at least years from date of receipt at -20° C. Upon reconstitution, this cytokine can be stored in working aliquots at2° -8° C for one month, or at -20° C for six months, with a carrier protein without detectable loss of activity. Avoid repeated freeze/thaw cycles.
Usage
This cytokine product is for research purposes only.It may not be used for therapeutics or diagnostic purposes.
Interactor
P17181
Interactor
Biological Process
Molecular function
Methods
Cytokine treatment
- After 2 passages, cells were seeded into 6-well plates .
- At confluence, cytokine treatment was performed during 48 hours using human recombinant TNF-α 100 U/ml or IFN-γ 200 U/ml in serum-free media, otherwise as above.
- Experiments were ended by removal and freezing of the supernatants and addition of lysis buffer to the cell monolayer, see below.
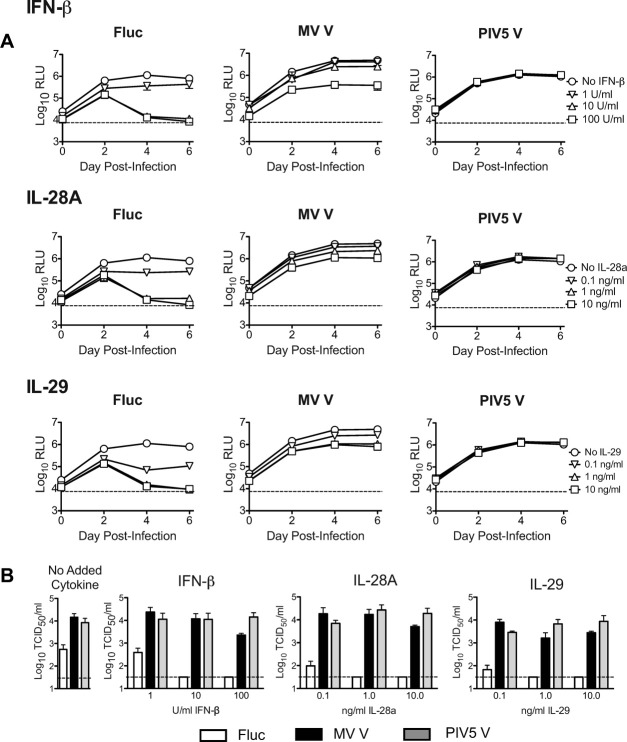
V protein expression counteracts the HCV-inhibitory effects of added type I and type III interferons in HFLC.
- Cells were washed three times then refed with HDM containing serial 10-fold dilutions of IFN-β, IL-28A, or IL-29.
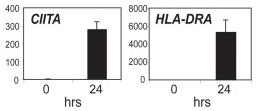
IFN-γ treatment induced the deposition of active histone modifications throughout the HLA-DRA gene.
- Transcript levels for A431 cells are shown before and after treatment with 500 U/ml of IFN-γ for 24 hrs.
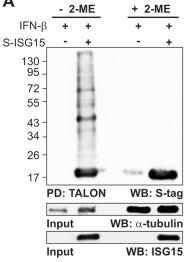
Decrease of ISG15 conjugates by reducing agents.
- 24 h post-transfection, the cells were induced with IFN-β (1,000 units/ml).
Cell Culture and Transfection
- HeLa B cells (ECACC # 85060701) were cultured in Dulbecco’s modified Eagle’s medium (Gibco) supplemented with 10% heat-inactivated calf serum (Sigma-Aldrich), penicillin (100 units/ml) (Gibco), streptomycin (100 µg/ml) and Non-Essential Amino Acids Solution 10 mM (100x) (Gibco) at 37°C with 5% CO2 in a humidified incubator.
- Cells were transiently transfected with GeneJuice according to the manufacturer’s instructions.
- For co-transfections, the total plasmid amount for each transfection was normalized by the addition of appropriate amount of empty vectors.
- For cytokine stimulation, 1,000 units/ml of human IFN-β was used.
Materials
- IFN-α and -β were from PBL Medical Laboratories (Piscataway, ), and IFN-λ1 (interleukin-29) was from .
- TNF-α was from Prospec-Tany .
- Interleukin-1β (IL-1β) and IFN-γ were from & .
- All of the cytokines were produced by recombinant expression of human genes.
- The functional activities of the IFNs were confirmed by demonstrating their abilities to induce IFN signaling, as indicated by translocation of STAT2 from the cytoplasm to the nuclei of treated cells (
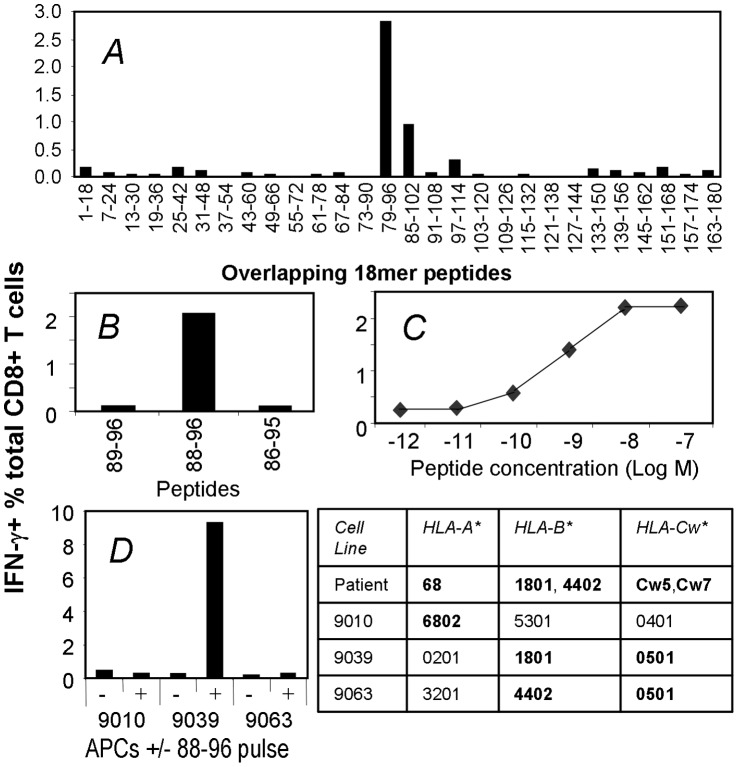
Identification and characterization of a novel NY-ESO-1 TCD8+ epitope.
- These cells were cultured with a panel of overlapping NY-ESO-1 18 mer peptides and then tested for responsiveness to each peptide in an ICS assay for IFN-γ.
CysLT1 receptor expression in human coronary artery smooth muscle cells is upregulated by pro-inflammatory stimuli.
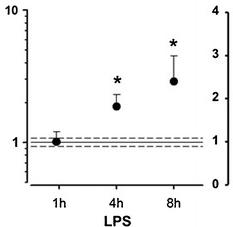
- Real-time quantitative TaqMan RT-PCR for CysLT receptor mRNA in SMCs incubated in the absence and presence of LPS (10 μg/mL) for 1, 4, and 8 h (a) and IL-6 (20 ng/mL), TNF-α (10 ng/mL), or IFN-γ (20 ng/mL) for either 8 h (b) or 24 h (c).
2. Generation of CIK Cells
- Human peripheral blood mononuclear cells (PBMCs) were obtained from health donors with written informed consent from all persons according to protocols approved by the Institutional Review Board of the Southwest Hospital, Third Military Medical University.
- CIK cells were generated as described previously
µ g/ml streptomycin. - Human recombinant IFN-γ were added at 1000 U/ml on day 0.
- After 24 hours of incubation, 50 ng/ml of antibody against CD3 (eBioscience, ), 100 U/ml IL-1α and 300 U/ml IL-2 were added.
- Cells were incubated at 37°C in a humidified atmosphere of 5% CO2 and subcultured every 3 days in fresh complete medium and 300 U/ml IL-2 at 3×106 cells/ml.
- The cells were cultured until 14 days to obtain CIK cells.
Infection protocol
- All infections were performed with CHIKV clone #4 at a MOI of 1.
- First, to test the susceptibility of the fibroblast cell lines HS 633T and HT-1080, cells were either grown on glass coverslips in 24-well plates or in Petri dishes until 60% of confluence and then incubated with CHIKV for different periods (8, 24 and 48 h PI).
- Mock-infected cells were prepared as a control.
- To compare the expression profile of HS 633T and HT-1080 innate immune genes, cells were grown in Petri dishes and infected with CHIKV for the same periods as above.
- Finally, to analyze the nuclear translocation of pSTAT1 in HS 633T and HT-1080, cells were grown on glass coverslips in 24-well plates until 40% of confluence and were either incubated with recombinant human IFN-α alone for 30 min at a final concentration of 100 ng/mL, infected with CHIKV for 24 h and unstimulated or infected with CHIKV for…